Analysis of Formation and Cracking of Phosphorus Segregation in Carbon Structural Steel
At present, the common specifications of carbon structural steel wire rods and bars provided by domestic steel mills are φ5.5-φ45, and the more mature range is φ6.5-φ30. There are many quality accidents caused by phosphorus segregation in small-sized wire rod and bar raw materials. Let's talk about the influence of phosphorus segregation and the analysis of the formation of cracks for your reference.
The addition of phosphorus to iron can correspondingly close the austenite phase region in the iron-carbon phase diagram. Therefore, the distance between the solidus and the liquidus must be enlarged. When phosphorus-containing steel is cooled from liquid to solid, it needs to pass through a wide temperature range. The diffusion rate of phosphorus in steel is slow. At this time, molten iron with a high phosphorus concentration (low melting point) is filled in the gaps between the first solidified dendrites, thereby forming phosphorus segregation.
In the cold heading or cold extrusion process, cracked products are often seen. The metallographic inspection and analysis of the cracked products shows that the ferrite and pearlite are distributed in bands, and a strip of white iron can be clearly seen in the matrix. In the ferrite, there are intermittent band-shaped light gray sulfide inclusions on this band-shaped ferrite matrix. This band-shaped structure caused by the segregation of sulfur phosphide is called "ghost line". This is because the phosphorus-rich zone in the area with severe phosphorus segregation appears white and bright. Due to the high phosphorus content of the white and bright belt, the carbon content in the phosphorus-enriched white and bright belt is reduced or the carbon content is very small. In this way, the columnar crystals of the continuous casting slab develop toward the center during the continuous casting of the phosphorus-enriched belt. . When the billet is solidified, austenite dendrites are first precipitated from the molten steel. The phosphorus and sulfur contained in these dendrites are reduced, but the final solidified molten steel is rich in phosphorus and sulfur impurity elements, which solidify in Between the dendrite axis, due to the high content of phosphorus and sulfur, sulfur will form sulfide, and phosphorus will be dissolved in the matrix. It is not easy to diffuse and has the effect of discharging carbon. Carbon cannot be melted in, so around the phosphorus solid solution (The sides of the ferrite white band) have a higher carbon content. Carbon element on both sides of the ferrite belt, that is, on both sides of the phosphorus-enriched area, respectively form a narrow, intermittent pearlite belt parallel to the ferrite white belt, and the adjacent normal tissue Separate. When the billet is heated and pressed, the shafts will extend along the rolling processing direction. It is precisely because the ferrite band contains high phosphorus, that is, the serious phosphorus segregation leads to the formation of a serious broad and bright ferrite band structure, with obvious iron There are light gray strips of sulfide in the broad and bright band of the element body. This phosphorus-rich ferrite band with long strips of sulfide is what we commonly call the "ghost line" organization (see Figure 1-2).

Figure 1 Ghost wire in carbon steel SWRCH35K 200X
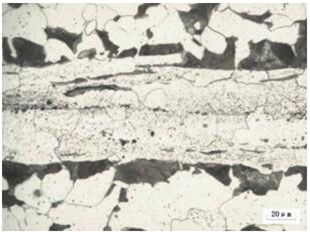
Figure 2 Ghost wire in plain carbon steel Q235 500X
When steel is hot rolled, as long as there is phosphorus segregation in the billet, it is impossible to obtain a uniform microstructure. Moreover, due to severe phosphorus segregation, a "ghost wire" structure has been formed, which will inevitably reduce the mechanical properties of the material. .
The segregation of phosphorus in carbon steel is common, but the degree is different. When the phosphorus is severely segregated (the "ghost line" structure appears), it will bring extremely adverse effects to the steel. Obviously, the severe segregation of phosphorus is the culprit of material cracking during the cold heading process. Because different grains in steel have different phosphorus content, the material has different strength and hardness; on the other hand, it is also Make the material produce internal stress, it will promote the material to be prone to internal cracking. In the material with "ghost wire" structure, it is precisely the reduction of hardness, strength, elongation after fracture and reduction of area, especially the reduction of impact toughness, which will lead to the cold brittleness of the material, so the phosphorus content and the structural properties of steel Have a very close relationship.
Metallographic detection In the "ghost line" tissue in the center of the field of view, there are a large number of light gray elongated sulfides. The non-metallic inclusions in structural steel mainly exist in the form of oxides and sulfides. According to GB/T10561-2005 "Standard Grading Chart Microscopic Inspection Method for the Content of Non-metallic Inclusions in Steel", the Type B inclusions are vulcanized at this time The material level reaches 2.5 and above. As we all know, non-metallic inclusions are potential sources of cracks. Their existence will seriously damage the continuity and compactness of the steel microstructure, and greatly reduce the intergranular strength of steel. It is inferred from this that the presence of sulfides in the "ghost line" of the internal structure of the steel is the most likely location for cracking. Therefore, cold forging cracks and heat treatment quenching cracks in a large number of fastener production sites are caused by a large number of light gray slender sulfides. The appearance of such bad weaves destroys the continuity of metal properties and increases the risk of heat treatment. The "ghost thread" cannot be removed by normalizing, etc., and impurity elements should be strictly controlled from the smelting process or before the raw materials enter the factory.
Non-metallic inclusions are divided into alumina (type A) silicate (type C) and spherical oxide (type D) according to their composition and deformability. Their existence cuts off the continuity of the metal, and pits or cracks are formed after peeling. It is very easy to form a source of cracks during cold upsetting and cause stress concentration during heat treatment, resulting in quenching cracking. Therefore, non-metallic inclusions must be strictly controlled. The current steel GB/T700-2006 "Carbon Structural Steel" and GB/T699-2016 "High-quality Carbon Structural Steel" standards do not make clear requirements for non-metallic inclusions. . For important parts, the coarse and fine lines of A, B, and C are generally not more than 1.5, and the D and Ds coarse and fine lines are not more than 2.
Post time: Oct-21-2021